Activating Amgen: A map for Selling to Amgen
March 02, 2020 | Xhermi Trimi |
Industry Articles
Amgen Inc. is one of the world’s 10 largest biopharmaceutical companies, founded and headquartered in California, USA. In 2018, Amgen ranked 10th on Pharmaceutical Executive’s list of top 50 highest-grossing global pharmaceutical companies [1]. The company’s prescription sales in 2017 came up to about $21.8 billion USD.
Amgen is a relatively young company, initially founded in 1980 under the name Applied Molecular Genetics Inc. (shortened to Amgen in 1983). After years of tireless research, their research team isolated and cloned the human erythropoietin gene in 1983. This allowed them to create biotech’s first blockbuster drug, epoetin alfa, which received FDA approval in 1989. Building on this initial success, the company has developed several other blockbuster drugs since then.
Business Overview
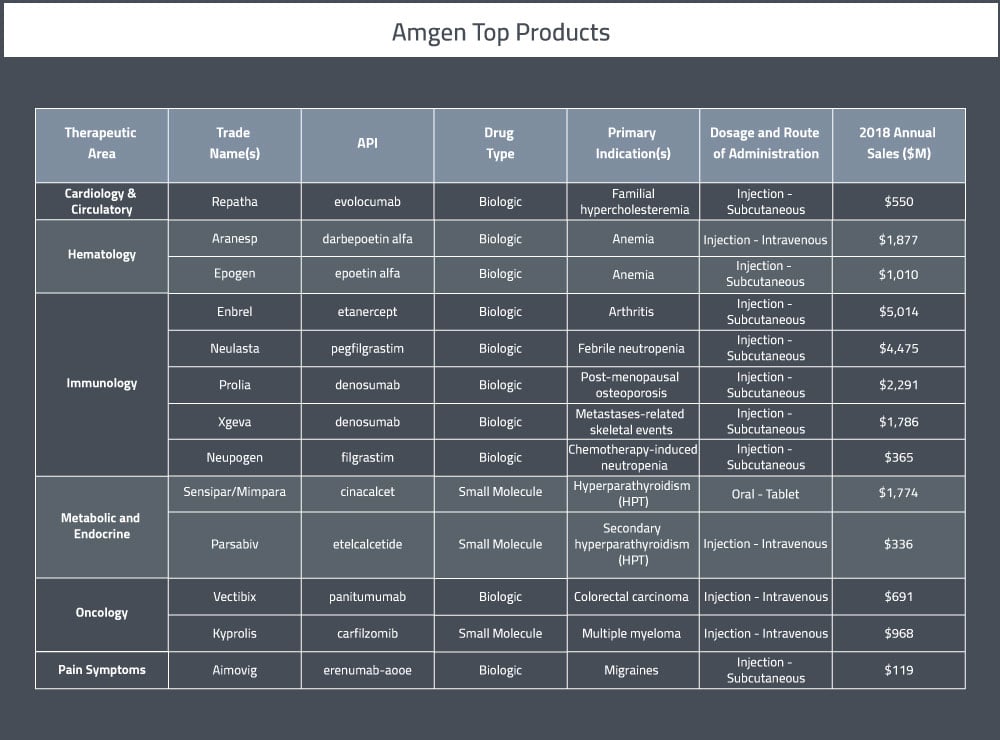
At the end of 2017, Amgen had over 20,000 employees in approximately 100 countries worldwide [2]. Their primary therapeutic areas of focus are oncology, hematology, cardiovascular disease, inflammation, bone health, nephrology and neuroscience. A summary of their top pharmaceutical products can be found in Table 1 below.
In 2017, Amgen’s R&D expenditures totalled around $3.5 billion USD, down about 8% compared to 2016 [2]. The company’s strategic priorities for R&D are: Innovative medicines, global geographic reach, next-generation bio-manufacturing, improved drug delivery systems and branded biosimilars. In their pipeline, they currently have twenty-two Phase I candidates, three Phase II candidates and seven Phase III candidates. Additionally, they are currently developing biosimilars to infliximab, rituximab and eculizumab. There are currently 257 active clinical trials being sponsored by Amgen.
Outsourcing History
Amgen regularly outsources various functions to third party companies. This includes product manufacturing functions, clinical trial product manufacturing, raw material sourcing, and distribution. It also includes clinical trial activities and even research and development collaborations.
In geographic areas where they do not have their own facilities and/or where it allows them to strategically mitigate potential supply impacts and risks; Amgen uses third party contract manufacturers for various manufacturing functions (such as: bulk manufacturing, formulation, fill, finish and device assembly) [2]. They also use third party distributors to supplement distribution worldwide [2]. Recently, in March 2016, they entered into a “flexible manufacturing agreement” with Patheon [3].
Amgen’s clinical trial products are mainly produced in their American facilities, but they also occasionally contract third party manufacturers for production [2]. Their clinical trial activities are conducted by both their internal staff and by third party contract clinical trial service providers [2].
Certain raw materials, medical devices (e.g. companion diagnostics) and components for manufacturing are provided by unaffiliated third party suppliers. For example, in December 2017, they entered into an agreement with Biocartis to help them develop their RAS biomarker test, which will be used as a companion diagnostic for Amgen’s colorectal cancer drug, Vectibix [4]. They entered into an exclusive agreement with Unilife in January 2016 for the production of a wearable injector device [5].
Amgen also has a history of entering into research collaborations with various types of partners. In 2016, CRO Syngene International began construction of a R&D Center for Amgen in Bangalore, India [6]. In 2018, they entered into a strategic research collaboration with Entera Bio to use their proprietary drug delivery platform in order to develop oral formulations for one of their preclinical inflammatory disease candidates [7].
Have a Zymewire account? Click here to view Amgen’s global activity!
Drug Development Sites
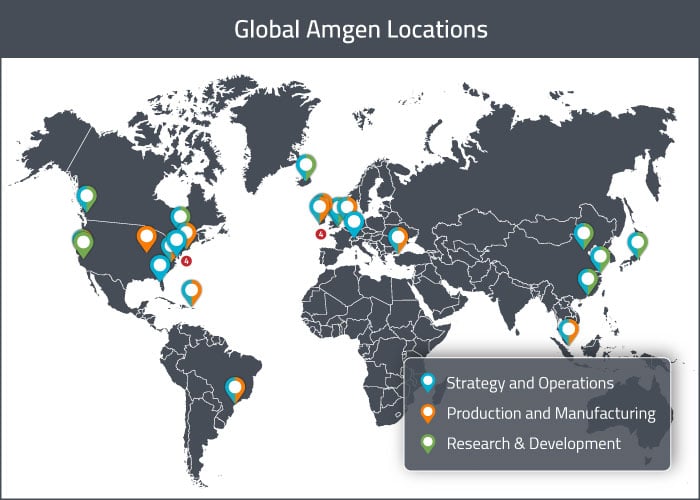
At the end of 2017, Amgen owned or leased about 180 different properties internationally [2]. Their corporate headquarters is located in Thousand Oaks (California), on a campus that houses manufacturing facilities, administrative offices, R&D facilities, sales and marketing offices, warehouses and distribution centers. Their main locations internationally are summarized in the map below and described in greater detail in the following sections.

Research sites
The company has major R&D centers in the Americas in Thousand Oaks (California), San Francisco (California) and Cambridge (Massachusetts). In Europe, they also have major R&D centers in Iceland and the United Kingdom. Other smaller research centers and development facilities also exist elsewhere globally.
Manufacturing Sites
Amgen manufactures both biologics and small molecule drugs, but the majority of their products are biologics which must be produced in living cells. Internally, they possess bulk manufacturing, formulation, fill, finish and device assembly facilities.
For the Americas, these activities are performed in the United States (in 4 different states), in Puerto Rico and in Brazil. For Europe, facilities exist in the Netherlands, Ireland and Turkey. For APAC, there is a facility in Singapore; a second facility in Singapore is currently being qualified. The Singapore facility is fully reconfigurable, allowing for the production of other products for worldwide supply.
The American manufacturing sites and the Netherlands site also perform the majority of global manufacturing support functions such as quality control, process development, procurement, production scheduling and warehousing. The company also uses third party contract manufacturers to develop similar capabilities in multiple other geographic areas; this allows the mitigation of potential supply impacts and risks. Clinical trial products are mainly produced in the American facilities or by third party manufacturers.
Distribution Centers
Amgen has distribution centers in Kentucky (California) and Netherlands for worldwide distribution of the majority of their commercial and clinical products, but they also use third party distributors to supplement distribution worldwide.
Have a Zymewire account? Click here to view Amgen’s global activity!
Decision Making
Decision making has been classified into three sections: clinical outsourcing, manufacturing and logistics. This is to facilitate outreach efforts by different types of companies looking to sell to or partner with this company.
Where are their clinical outsourcing decisions based out of?
The general trend encompassing clinical decision is that they are based out of the Greater Los Angeles Area for the Americas and more dispersed for Europe and APAC.
For the Americas, you’ll find titles like Outsourcing Manager, Global Regulatory Outsourcing Specialist, Senior Outsourcing Manager, Clinical Manager, Director Global Clinical Management, US Head of Clinical Research and Director Clinical Development in the Greater Los Angeles Area. You’ll also find a Director of Research in the Greater Boston Area and a multiple Clinical Trial Managers based out of Toronto (Ontario, Canada).
For Europe, there are several clinical outsourcing decision makers in the United Kingdom. Specifically, you should look out for titles like Director of Global Clinical Management and Director of Clinical Research in Cambridge and Uxbridge. Elsewhere in the UK (such as Sheffield, Southall, London, Leeds, etc.), there are multiple Clinical Trial Managers. Clinical Trial Managers are also found in France (Lyon and Paris), Belgium (Antwerp and Brussels), Spain (Barcelona), Germany (Munich) and the Netherlands (Breda Area). Finally, a Director of Clinical Research is located in Paris, France.
For APAC, there are Clinical Study Managers in China (Shanghai) and Australia (Sydney). There are also Clinical Operations Managers in Australia (Melbourne and Brisbane) and India (Mumbai). Finally, there is a Director of Discovery Research in China (Shanghai) and a Director of Clinical Research in Australia (Sydney).
Where are their manufacturing decisions based out of?
Manufacturing decisions seem to be based mostly out of Puerto Rico, Rhode Island, and the Greater Los Angeles Area for the Americas, mainly out of Ireland for Europe, and exclusively out of Singapore for APAC.
In the Americas, you will find multiple Manufacturing Directors and Manufacturing Managers. These are the two main titles to look out for. You’ll also find Manufacturing Directors and Manufacturing Managers in Puerto Rico, the Greater Los Angeles Area (California, USA), and the Greater Boston Area (Massachusetts, USA).
In Europe, Manufacturing Managers are located in Ireland (Leinster), Germany (Munich) and Belgium (Antwerp Area). There is a Contract Manufacturing Manager and a Director of Manufacturing in Leinster, Ireland. In the Netherlands, you’ll find multiple Manufacturing Managers in the Groningen and Breda Areas. Finally, you’ll find another Contract Manufacturing Manager in the Zurich Area of Switzerland.
In APAC, Singapore is where manufacturing decisions are based out of. There you’ll find multiple Manufacturing Managers, you’ll also find a Manufacturing Director.
Where are their logistics operations based out of?
Logistics decision are pretty dispersed in each of the three markets. In the Americas, they seem to be split across Puerto Rico, the Greater Los Angeles Area, and Toronto. For Europe; Netherlands, Ireland, Turkey, Belgium and Germany seem to be the main spots for logistics decision makers. For APAC, they’re split across Singapore, Australia, China and India.
For the Americas, Supply Chain Manager is a title to look out for—you’ll find multiple decision makers with this title in Toronto (Ontario, Canada), the Greater Los Angeles Area (California, USA), and Puerto Rico. In Puerto Rico, there are also multiple Supply Chain Directors. Finally, there are several Procurement Managers in Tampa (Florida, USA), St. Petersburg (Florida, USA), and the Greater Los Angeles Area (California, USA).
For Europe, you’ll find Supply Chain Managers and Supply Chain Directors in the Netherlands (Breda Area), Turkey (Istanbul), and Ireland (Leinster and Dublin). There is also a Procurement Manager in Istanbul, Turkey. You’ll find the Head of Sourcing and Procurement in Munich, Germany.
For APAC, you will once again want to keep an eye out for Supply Chain Directors, which you will find in Singapore (Singapore) and China (Beijing). You’ll also want to keep an eye out for the familiar title of Supply Chain Manager in Singapore (Singapore), Australia (Sydney and Melbourne) and India (Bengaluru Area). There is also a Supply Chain Head located in the Hyderabad Area of India.
Have a Zymewire account? Click here to view Amgen’s global activity!
Innovation
One way Amgen fosters innovation is through Amgen Ventures. Amgen Ventures provides resources to innovative biotechnology start-up companies to assist them in developing promising new therapeutics. Since it’s inception, Amgen Ventures has helped over 30 companies. They invest in opportunities across North America and Europe [8].
Amgen has also played a key role in establishing life sciences innovation hubs. In September 2018, Amgen entered into an agreement with the County of Los Angeles to become a founding sponsor of BioLA. This newly formed organization is focused on creating a new life science innovation hub in Los Angeles County; bringing together companies, academic institutions, hospitals, investors, trade associations and public agencies [9]. They were also a pioneer in setting up the innovation hub in Cambridge (Massachusetts), starting in the area 25 years ago [10].
In Europe, Amgen also fosters innovation through innovation hubs, just as their Technology Hub in Berlin. In October 2017, Amgen entered into a partnership with EIT Health via a meeting at their Berlin Technology Hub [11]. EIT Health is a network of over 140 health innovators (pharma/medtech companies, investors, research institutions and universities) that is backed by the European Union.





.png?width=500&name=Q3%202024%20Biopharma%20Recap%20(Zymewire).png)

Comments